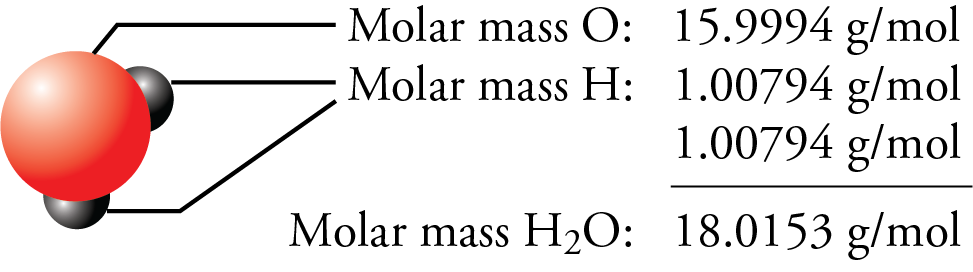
The factor-label method yields the desired cancellation of units, and the computed result is on the order of 10 22 as expected. The term mole is defined in that one mole of a substance with a molecular (or atomic) mass of one (1), will have a mass of 1 gram. For KMnO4: Molar mass of K 39.1 g Molar mass of Mn 54.9 g Molar mass of O 16.0 g Molar mass of KMnO4 39.1 g + 54.9 g + (16.0 g x 4) Molar mass of KMnO4 158. From the periodic table the molar masses of the compounds will be extracted. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6. To convert grams to moles, the molecular weight of the solute is needed. One mole of a substance has the same mass in grams that one atom or molecule has in atomic mass units.\] The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. Therefore, we must use the molar mass of calcium from the periodic table (40.08 g/mol) to convert grams to moles of calcium. The numbers in the periodic table that we identified as the atomic masses of the atoms not only tell us the mass of one atom in u but also tell us the mass of 1 mol of atoms in grams. 
One mole of a substance has the same mass in grams that one atom or molecule has in atomic mass units. The atomic masses can be found on a periodic table. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of Na atoms has an approximate mass of 23 grams. The molar mass is determined by summing the atomic masses of each element in the molecular formula. Substitute the known values: mass 70.906 g/mol × 0.560 mol 39. Use the molar mass formula to calculate the mass: mass molar mass × number of moles. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of H atoms has a mass of approximately 1 gram. Since chlorine has a molar mass of 35.453 g/mol on the periodic table, the molar mass of the chlorine gas Cl 2 is twice this value. The molar mass is numerically the same as the atomic or molecular weight, but it has. MHg2Br2 mHg2Br2 nHg2Br2 560.98 g mol1 M Hg 2 Br 2 m Hg 2 Br 2 n Hg 2 Br 2 560.98 g mol 1. How many particles are there in 1.43 g of a molecular compound with a gram molecular mass of 233 g 6. For toluene that is: 12.01 7 (for Carbon) added to 1.009 8 for hydrogen. The atomic mass is the number of grams per mole of a substance. Add the atomic masses of the elements to find the mass of one mole. The atomic mass of hydrogen is 1.0079 and the atomic mass of oxygen is 15.9994. 
One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. Find the mass, in grams, of 1.00 x 1023 molecules of N2. Remember that in order to find the molar mass, you find the mass of each element and multiply by the number that exists within the molecule and add it all together. Look up the atomic masses of hydrogen and oxygen from the periodic table. Molar mass is the mass (in atomic mass units) of one mole of a of a substance. Determine the volume in liters occupied by 14 g of nitrogen gas at STP. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12. Find the mass in grams of 2.00 x 1023 molecules of F2. b) Similarly, for Hg 2 Br 2 the molecular weight is 560.98, and so. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. STP Gas relationship: at STP, 1 mole of gas 22.4 L gas. The number in a mole, Avogadro’s number, is related to the relative sizes of the atomic mass unit and gram mass units. a) The atomic weight of mercury is 200.59, and so 1 mol Hg weighs 200.59 g. Use atomic masses from the periodic table to calculate molar mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
